HOW TO USE CALCULATOR

3.3. Results and recipe
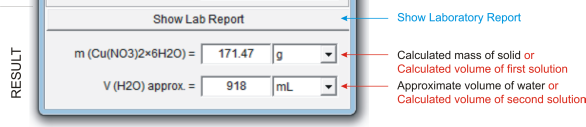
The first field displays the calculated mass of solid compound or the calculated volume of first solution. You can choose a unit in which the mass (volume) is expressed.
The second field displays the approximate volume of solvent (water) or the calculated volume of second solution. You can choose a unit in which the volume is expressed.
SolCalc automatically calculates the necessary amount of chemicals as soon as some information changes. The result will automatically convert itself when you chose a new unit of measurement from the drop down menu.
Unit conversions are an important part of solution calculations. You can convert from small SI units to large SI units or to small English Units, and so on. To view the complete list of the units, go to Unit conversion page.
Lab Report
By clicking on the Show Lab Report button you will get the complete list of all calculations that were done and recipes for preparation of the desired solution. From this new window, you can print the report or save it to your computer by clicking on the Print or Save button at the bottom of the window. If you choose to save the report, it will be saved as a txt file.
Available below is a couple of lab reports, to give you an example of what you can do with SolCalc.
- How to dilute a protein stock solution?
- Construct a calibration curve by plotting the mean absorbance for each standard concentration against the target protein concentration. The concentration of the stock solution is 100 mg/L, and the volume of the diluted sample is fixed at 2 mL. Protein standards should be prepared in the same buffer as the samples to be assayed. A convenient standard curve can be made using bovine serum albumin with concentrations of 0, 10, 20, 30, 40, 50 mg/L.
- How to mix two ammonia solutions?
- A chemist has one solution that is 28 % ammonia and another solution that is 10 % ammonia. How many gallons of each should be mixed to get 3 gallons of a solution that is 20 % ammonia?
- How to make 0.1 M hydrochloric acid?
- Calculate the volume of concentrated hydrochloric acid, having a density of 1181 g/L and containing 36.5 % HCl by weight, needed to prepare 500 mL of a 0.1 mol/L HCl solution?
- How to make a copper(II) sulfate solution?
- How many grams of copper(II) sulfate pentahydrate (CuSO4×5H2O) would you need to dissolve in water to produce 1 liter of a 20 % aqueous solution of copper(II) sulfate?
Citing this page:
Generalic, Eni. "SolCalc Help: Results and recipe." EniG. Periodic Table of the Elements. KTF-Split, 27 Oct. 2022. Web. {Date of access}. <https://www.periodni.com/enig/solcalc_help/results_and_recipe.html>.
Articles and tables
- Periodic table
- Online calculators
- Scientific calculator for chemists
- Gas laws calculator
- Molar mass calculator
- Angle converter
- Roman numerals converter
- Number systems converter
- Preparation of solutions
- Labeling of chemical containers
- Oxidation numbers calculator
- ARS method
- Oxidation number change method
- Ion-electron method
- Gauss elimination method
- Memory game
- Find the pairs
- Articles and tables
- Chemistry
- List of abbreviations and acronyms
- Crystal systems and Bravais lattices
- GHS - Hazard pictograms
- NFPA 704 Hazard Diamond
- Fundamental physical constants
- Solubility product constants
- SI - International System of Units
- Composition of mixtures and solutions
- Stoichiometric calculations
- Chlorinity and salinity of seawater
- Rare earth elements (REE)
- Ecology
- Web design
- Chemistry dictionary
- Chemistry
- Downloads
- ≡ Menu
